
| Strona główna |
| Zespół |
| Badania |
| Aparatura |
| Seminaria |
| Publikacje |
|
Nasze konferencje |
|
Aktywność konferencyjna |
| Projekty |
| Programy |
|
Najbliższe wydarzenia |
| Linki |
| Kontakt |
|
|
|
od 2020-09-20 |

Authors: Niziński S., Popenda Ł., Rode M.F., Kumorkiewicz A., Fojud Z., Paluch-Lubawa E., Wybraniec S., Burdziński G. |
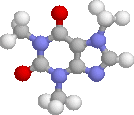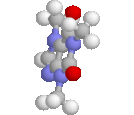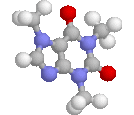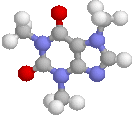
Title: Structural studies on the stereoisomerism of a natural dye miraxanthin I |
Source: New Journal of Chemistry |
Year : 2019 |
The chemical structure of a yellow dye present in Mirabilis jalapa L., miraxanthin I, was characterized by NMR spectroscopy. The extract of M. jalapa was analysed by a high-performance liquid chromatographic system (LC-DAD-FL-ESI-MS/MS) and the presence of miraxanthin I among other betaxanthins was confirmed. This dye was synthesized from previously generated methionine-betaxanthin by oxidation with 10% H2O2. The E/Z stereoisomers of miraxanthin I were found by NMR analysis to occur in a 50 : 33 : 10 : 7 ratio in aqueous solution (7E,9E : 7Z,9E : 7E,9Z : 7Z,9Z configurations, respectively). Comparison of NMR data with chemical shifts obtained from quantum chemical calculations strongly suggests the presence of intramolecular hydrogen bonds which may favour a more rigid structure of the dye. This explains the highest fluorescence quantum yield among betaxanthins. The observed changes in the H-1 NMR spectra during the measurements indicate on miraxanthin I hydrolysis for which a mechanism is proposed. The first step during the hydrolysis process is protonation of the nitrogen atom within the central bridge bond system which is observed in the ZE stereoisomer by NMR. |
|
|
Zaktualizowano: podstrony 2021-06-21 / bazę danych: 2024-11-02 by Webmaster: Zbigniew Fojud